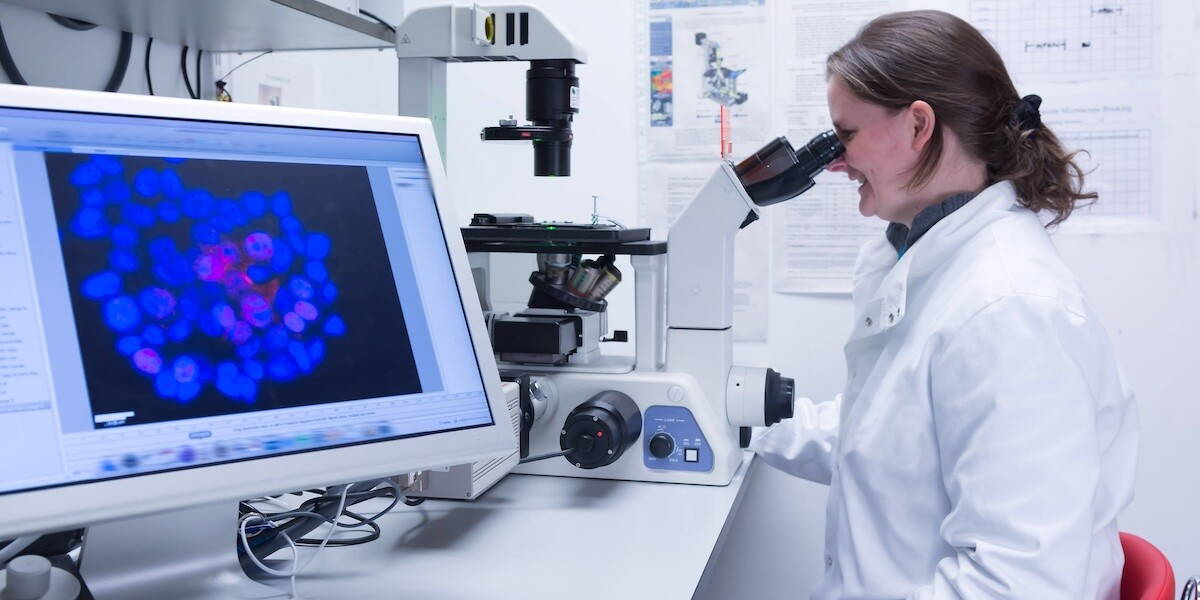
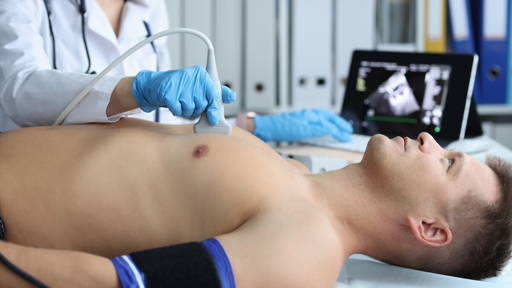
Researchers at Oregon Health & Science University have developed a promising blood test that could significantly improve early detection of pancreatic cancer, one of the deadliest forms of cancer worldwide. The technique uses nanoparticles and a microchip-based electrical signal to identify cancer-specific biomarkers circulating in the blood.
Pancreatic cancer is notoriously difficult to detect at an early stage. Located deep within the body, tumors often go unnoticed until symptoms such as jaundice or abdominal pain appear, typically when the disease has already progressed. This late diagnosis contributes to poor survival rates and limited treatment options.
The newly developed approach aims to change that by enabling earlier, less invasive detection, particularly among high-risk populations such as individuals with a family history of the disease. The findings were described in the journal Small.
How the technology works
The innovation centers on a microchip that delivers a small electrical pulse to isolate nanoparticles released by tumors into the bloodstream. These particles carry molecular signatures, biomarkers, that can indicate the presence of cancer.
Once captured, the nanoparticles are treated with fluorescent markers that highlight these cancer-related signals. According to lead researcher Stuart Ibsen, the intensity of the signal increases with the number of biomarkers present. “The more cancer biomarkers, the brighter the electrodes on the chip become,” he explained.
Because tumors shed large quantities of these nanoparticles, they can be detected through a relatively simple blood draw. This makes the method far less invasive than traditional diagnostic approaches, such as tissue biopsies.
Promising results
In collaboration with the Brenden-Colson Center for Pancreatic Care, the research team conducted an early-stage, blinded study involving blood samples from 36 participants. The cohort included patients diagnosed with pancreatic cancer as well as individuals with noncancerous pancreatic conditions, such as pancreatitis.
The results were striking. The test demonstrated a 97 percent accuracy rate in distinguishing cancerous cases from benign conditions, thus significantly outperforming conventional biopsy techniques. Standard biopsy procedures, which involve ultrasound-guided needle extraction of tissue, typically detect around 79 percent of pancreatic cancers.
Importantly, the new method was also able to differentiate between malignant tumors and benign or precancerous lesions. This capability could have major clinical implications, helping physicians avoid unnecessary surgical interventions for patients with noncancerous growths.
Clinical application
While the findings are encouraging, the technology is still in an early phase of development. Researchers estimate that it may take up to five years before the test becomes available for routine clinical use.
If validated in larger trials, the approach could transform the diagnostic pathway for pancreatic cancer. A minimally invasive blood test that reliably detects early-stage disease would not only improve survival outcomes but also reduce the burden on patients and healthcare systems.
Beyond pancreatic cancer, the underlying technology may also have broader applications in oncology. By targeting tumor-derived nanoparticles, similar platforms could potentially be adapted to detect other hard-to-diagnose cancers.
For now, the study highlights the growing role of nanotechnology and microchip-based diagnostics in advancing precision medicine, offering a glimpse into a future where early detection becomes the norm rather than the exception.